POOR QUALITY EMBRYOS IMPLANTATION POTENTIAL
Cristina Torrado, Marta Guijarro, María M. Hebles, Mónica Dorado, Javier Ávila-Medina, Pascual Sánchez.
aClínicas Ginemed Sevilla. Farmacéutico Murillo Herrera, 3. 41010, Seville. Spain; bClínicas Ginemed Huelva. Calle Punta Umbría, 8. 21002 Huelva, Spain.
The aim of our study was to determine whether extended culture of surplus embryos that are not cryopreserved on day 3 because of not enough quality according to ASEBIR embryo grading, are capable to reach blastocyst stage, with optimum quality to be cryopreserved and transferred if needed during the following cycle.
1462 stimulated cycles performed in “Clínicas Ginemed” in 2016 from 1372 patients were analysed in a retrospective study. Best quality embryo/s were transferred on day 3 and supernumerary good quality ones were vitrified. Blastocysts originated from poor quality embryos (graded as D on day 3) were vitrified only if trophectoderm and inner cell mass were graded between A and C.
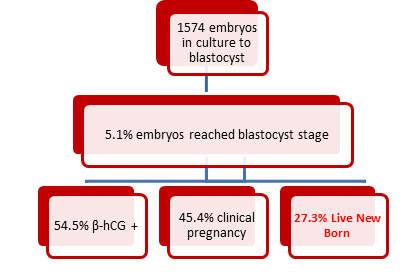
5.1% of all poor quality embryos obtained from 1462 stimulated cycles reached blastocyst, of those 46.25% were graded as B and 53.75% as C, and vitrified ;being 11 of them transferred after 100% warming survival, reaching an ongoing pregnancy on 45.4% cases (5 out of 11) of which 2 miscarriages between the seventh and eight week. And the final new born rate were 27.3%.
Surplus embryos that would be discarded following morphological criteria could have a second chance of being transferred if they were grown until day 5 or 6 of embryo culture, developing and implanting with a frequency reasonable of baby born with embryos who could be discarded in early developing stage.
INTRODUCTION
Outcomes in assisted reproduction techniques depend mainly in a proper embryo selection for transfer.
When cleavage stage embryos are transferred (second or third day of culture), embryo morphology is the most predictive factor regarding implantation (Puissant et al., 1987; Shulman et al., 1993; Ziebe et al., 1997; Balaban et al., 2001). Top quality embryos, according to Spanish Association for the Study of Reproductive Biology (ASEBIR in its Spanish initials) updated embryo classification (Ardoy et al., 2008; Mantilla et al., 2015), have been determined to have a higher implantation potential compared to lower graded quality embryos (based on division patterns, fragmentation, multinucleation and symmetry).
A common strategy to improve implantation rates when only poor quality embryos are available for transfer, is to increase the number of embryos to be replaced. However, a big variability is observed between patients when transferring similar quality embryos: some cases results in implantation failure and in others multiple pregnancy is observed (Ryan et al., 2007; Styer et al., 2008; Guerif et al., 2011; Shaw-Jackson et al., 2013).
The day of transfer is a decision that depends on the clinician, the patients, and policies among different clinics, and so the number of embryos to be transferred. The current advances in techniques applied on reproductive laboratories, have improved the conditions for extended embryo culture together with the chance to get good quality blastocysts. Current studies confirm that transferring good quality blastocysts is associated with a higher implantation potential (Balaban et al., 2001; Milki et al., 2002),.The main reason is that blastocysts have reached a step forward in development compared to cleavage stage embryos and that reducing the number of embryos replaced to uterus, and so the number of multiple pregnancies and associated risks.
Although most of reproductive clinics have similar criteria regarding embryo selection for transfer, there is still a lack of consensus in the embryos selected for cryopreservation. When a reproductive laboratory follows very strict selection criteria, they might be discarding embryos with a reasonable implantation potential. Several studies have demonstrated that poor quality embryos have potential to develop to blastocyst and are able to implant (Styer et al., 2008; Shaw-Jackson et al., 2013; Kaartinen et al., 2015).
The protocol stablished in “Clinics Ginemed” in 2016 consisted on a fresh embryo transfer of good quality embryos on day 3 of development, classified A or B according ASEBIR criteria (Ardoy et al., 2008; Mantilla et al., 2015), cryopreservation of similar quality supernumerary embryos and/or culture to blastocyst stage of poor quality surplus embryos, graded as D agreeing to ASEBIR. Those able to reach blastocyst stage on day 5 or 6 will be cryopreserved for future transfer.
The aim of this study is to evaluate whether extended culture of surplus embryos that are not cryopreserved on Day 3 because of not enough quality according to ASEBIR embryo grading, are capable to reach blastocyst stage, with optimum quality to be cryopreserved and transferred if needed during the following cycle.
MATERIAL AND METHODS
Inclusion criteria
Retrospective descriptive study of a total of 1462 cycles from 1372 patients performed in “Clinicas Ginemed”between January 2015 to September 2016. All patients included had a fresh embryo transfer on day 3 and had both, good quality embryos for vitrification and poor quality embryos to be cultured to blastocyst.
Controlled Ovarian Stimulation and oocyte pick up
Five days after finishing contraceptives, stimulation begins with 150-200 IU daily of r-FSH (alpha follitropin, Gonal-F, Merck, Spain) and 75 IU hMG (Menopur 75, Ferring, USA), depending on body mass index. After day five, determination of estradiol levels and ultrasound scans are performed for dose adjustment. When a follicle reached 14 mm, GnRH antagonist treatment with 0.25 mg Orgalutran (Organon, Ireland) is started until at least three follicles reached 20 mm. At this point, ovulation is triggered with 250 µg recombinant human Chorionic Gonadotropin (r-hCG) (Ovitrelle, D Merck, Spain).
Oocyte retrieval is performed 34-36 hours after hCG trigger injection by eco-guided ovarian puncture. A vaginal ultrasound transducer is used to visualize the ovary and follicles, and a single lumen ovum aspiration needle (Cook Medical, IN, USA) is inserted in the transducer and advanced into the ovarian follicles, where oocytes are aspirated with a syringe, until all the follicles are punctured. Then the oocyte-cumulus complexes are picked up under a microscope and placed in culture in gasified media droplets (25 µl) (G-IVF, Vitrolife, Sweden) under mineral oil, 6% CO2, and 37 ºC until denudation.
Embryo culture
All oocytes retrieved were denuded in Hyaluronidase solution (Hyase, Vitrolife, Sweden) cultured in gasified media droplets (25 µl) (G1-plus, Vitrolife, Sweden), under mineral oil, 6% CO2, and 37 ºC until day 3. Embryos cultured to blastocyst were transferred to G2-plus (Vitrolife, Swedwen) 25 µl droplets under low O2 tension conditions (6% CO2, 5% O2, 37 ºC).
Fertilization was assessed by observing two pronuclei and two polar bodies 17-20 hours after microinjection. Embryo development was assessed on days 2 and 3 with inverted microscope.
Best quality embryo/s, according to ASEBIR classification criteria (Ardoy et al., 2008; Mantilla et al., 2015), were transferred on day 3 and supernumerary good quality ones were vitrified. Embryos considered as poor quality to be cryopreserved, were placed on a new gasified media droplets (G2-plus, Vitrolife, Sweden) under mineral oil and cultured until day 5 or 6: embryos with ˃35% fragmentation (Munné, 2006), slow cleavage timing (6 cells or less) or fast cleavage embryos (Magli et al., 2007) were vitrified only if able to reach blastocyst stage as a final grading between A and C . However, the final decision whether lower quality embryos were vitrified or not was taken by the embryologist according to the center standardized protocols.
Vitrification/Warming Protocol
Vitrification was performed using the Cryotech Vitrification and Warming Kits (Cryotech, Japan), combined with Cryotech straws (Cryotech, Japan), according to manufacturer´s protocol (Kuwayama, 2012). Quality of the embryos was assessed after warming. Blastocysts with no damage, or partial damage were considered to have survived and were transferred.
Pregnancy confirmation
Confirmation of embryo implantation was performed by serum β-hCG hormone measurement 12 days after embryo transfer. Pregnancy was confirmed 4 weeks after embryo transfer by presence of intrauterine gestational sacs.
Statistical analysis
The statistical analysis consisted on a descriptive study of the analysed parameters on both groups: patients with fresh embryo transfer and those with embryos vitrified that reached blastocyst stage.
RESULTS
A total of 1462 stimulated In Vitro Fertilization (IVF) cycles with day 3 fresh embryo transfer were analysed. The characteristics data from all cycles were summarized in A1. Additionally, we obtained data form cycles with supernumerary embryos that were not transferred neither vitrified. Characteristics of those embryos are shown on Table A.2.
A total of 8917 were obtained on day 3. 3169 of those embryos were transferred after being graded between A and C according to ASEBIR criteria; 4096 were cryopreserved. 1574 embryos were graded as poor quality (D) and kept in culture until day 5-6 of development.
After 5 or 6 days of culture, 80 of the embryos in culture after fresh embryo transfer, corresponding to 48 cycles, reached blastocyst stage with quality enough to be vitrified (5.1%). 11 of those patients had a blastocyst frozen embryo transfer after negative pregnancy test on the fresh cycle. Those blastocysts were the unique ones cryopreserved for this patients in that cycle. Embryo survival rate after warming was 100% (Table A.2).
Pregnancy rate when transferring those remaining embryos in blastocyst stage was 54.5%, pregnancy was confirmed by measuring β-hCG in serum. In 66,6% of them from embryo transfered graded as B, and 0% the embryo transferred graded as C. Ongoing pregnancy was confirmed in 45.4% of cases after ultrasound. New born rate after transferring remaining embryos was 27.3%.
DISCUSSION
It is a common practice on reproductive clinics to transfer or cryopreserve good quality embryos, and to discard poor quality ones according to morphological criteria.
Our results show that embryos discarded when following morphological criteria stablished on most Assisted Reproductive clinics, not selected to be transferred or cryopreserved could continue their development to blastocyst stage and be cryopreserved on this stage. Those blastocysts showed good results after warming and transfer.
In this study, 80 blastocysts (5.1%) were vitrified out of 1574 embryos left in culture. 11 of them were transfered with 45.5% ongoing pregnancies. Baby at home rate was 27.3%.
Similar studies showed 6.6% blastocysts developed from poor quality embryos, with 40.9% implantation rate (Ren et al., 2012). Balaban concluded a high pregnancy rate by transferring blastocysts developed from poor quality embryos (Balaban et al., 2001). These studies show the limited value of embryo morphology as only parameter for embryo selection (Milki et al., 2002; Desai et al., 2014; Kaartinen et al., 2015), being useful in some cases other parameters.
By introducing new techniques in assisted reproductive laboratories, such as time-lapse, combined with metabolomic or proteomic studies (Scott and Treff, 2010; Desai et al., 2014), and together with morphological embryo grading can help to select the embryo with a better implantation potential but should not necessarily imply to discard those embryos that do not fit to these selection criteria.
Vitrification techniques allowed a significant improvement on embryo survival after warming. Data in our laboratory is in accordance with those described on literature (Osada et al., 2003; Stehlik et al., 2005; Edgar and Gook, 2012), achieving a 99% survival after warming is shown.
Some authors concluded that implantation rates after transferring blastocysts that survived to warming are similar to those presented by fresh blastocysts (Zhu et al., 2011). When embryos are cryopreserved on blastocyst stage, the higher number of cells might contribute to a better survival.
Our results show a high survival rate after warming blastocysts developed from day 3 poor quality embryos; they support the strategy presented, where any embryo is discarded on day 3, even when considered low quality embryos. Those embryos are cultured to blastocyst and if they reach the stage, they are vitrified. When warming the blastocyst for embryo transfer, the survival is close to 100% with good implantation rates. As a limitation of the current study, can be highlighted that every laboratory should study cost of extra culture against the number of patients to benefit of this strategy.
According with that it is interesting to think to keep embryo culture until blastocyst stage even in embryos graded as top quality on day 3. The ones who cames from poor embryo quality in day 3 can reach a blastocyst stage in 5.1% cases and the survivor rate after the vitrification process is close to 100%.To extend the embryo culture until day 5 and 6 is a no wasting cost and time in the laboratory, because most probably we might discard embryos who can have a fate in the future with a reasonable implantation potencial rate in every chance.
To conclude, according to data shown on this study, poor quality embryos can have a second chance to be cryopreserved and transferred afterwards if they are able to reach blastocyst stage, all of that with enough chances to survive warming and to implant. That was one of the reasons why we are including blastocyst culture in most of our cycles.
A. TABLES
| Value | SD | |
| Cycles with embryo(s) transferred | 1462 | |
| Mean age | 38.6 | 4.9 |
| MII/cycle | 8.4 | 3.2 |
| Number of embryos | 8917 | |
| Mean embryos/cycle | 6.1 | 2.7 |
| Embryos transferred | 3169 | |
| Mean fresh embryos transferred per transfer | 2.2 | 0.6 |
| % β-hCG positive value | 47.3 | |
| % Clinical pregnancies | 45.1 | |
| % Miscarriages | 15.2 | |
| % Baby at home | 37.7 | |
| Number of embryos cryopreserved | 4096 | |
| Number of embryos cryopreserved/cycle | 2.8 | 2.3 |
Table A.1. Characterictics of stimulated In Vitro Fertilization (IVF) cycles on day 3.
| Value | |
| Number of D3 surplus embryos cultured to blastocyst | 1574 |
| % Embryos cultured to blastocyst | 17.65 |
| Number of embryos cryopreserved (day 5/6) | 80 |
| % Surplus embryos cryopreserved | 5.1 |
| % Surplus embryos cryopreserved graded as B | 46.25 |
| % Surplus embryos cryopreserved graded as C | 53.75 |
| Number of cycles with only surplus cryopreserved embryos | 48 |
| Number of cycles with surplus warmed embryos transferred | 11 |
| % β-hCG positive value | 54.5 |
| % β-hCG positive value from Embryo grade B transfered | 66.7 |
| % β-hCG positive value from Embryo Grade C transfered | 0% |
| % Clinical pregnancies | 45.4 |
| % Miscarriages | 40 |
| % Baby at home | 27.3 |
Table A.2. Characteristics of stimulated In Vitro Fertilization (IVF) cycles on day 5/6 after transfer.
Fig. B.1. Schematic illustration showing the outcomes of the poor quality embryos which reached blastocyst stage.
Referencias
Ardoy M, Calderón G, Cuadros J, Figueroa MJ, Herrer R, Moreno JM, et al.. II Criterios ASEBIR de valoración morfológica de oocitos, embriones tempranos y blastocistos humanos. Cuad. Embriol. Clínica. Barcelona GÓBALO Gráfica 2008; 25–37.
Balaban B, Urman B, Alatas C, Mercan R, Aksoy S, Isiklar A. Blastocyst-stage transfer of poor-quality cleavage-stage embryos results in higher implantation rates. Fertil. Steril. 2001; 75:514–518.
Desai N, Ploskonka S, Goodman LR, Austin C, Goldberg J, Falcone T. Analysis of embryo morphokinetics, multinucleation and cleavage anomalies using continuous time-lapse monitoring in blastocyst transfer cycles. Reprod. Biol. Endocrinol. 2014; 12:54.
Edgar DH, Gook DA. A critical appraisal of cryopreservation (slow cooling versus vitrification) of human oocytes and embryos. Hum. Reprod. Update 2012; 18:536–554.
Guerif F, Frapsauce C, Chavez C, Cadoret V, Royere D. Treating women under 36 years old without top-quality embryos on day 2: a prospective study comparing double embryo transfer with single blastocyst transfer. Hum. Reprod. 2011; 26:775–781.
Kaartinen N, Das P, Kananen K, Huhtala H, Tinkanen H. Can repeated IVF–ICSI-cycles be avoided by using blastocysts developing from poor-quality cleavage stage embryos? Reprod. Biomed. Online 2015; 30:241–247.
Kuwayama M. The human oocyte: vitrification. Textb. Assist. Reprod. Tech. Fourth Ed. Vol. 1 Lab. Perspect. 2012; 285.
Magli MC, Gianaroli L, Ferraretti AP, Lappi M, Ruberti A, Farfalli V. Embryo morphology and development are dependent on the chromosomal complement. Fertil. Steril. 2007; 87:534–541.
Mantilla A, Orozco I, Zamora S, Ortiz N, Prados F, Moreno JM, et al.. Grupo de interés de calidad de ASEBIR: Actualización de las especificaciones para los indicadores de calidad de la Asociación para el Estudio de la Biología de la Reproducción (ASEBIR). Med. Reprod. y Embriol. Clínica 2015; 2:46–54.
Milki AA, Hinckley MD, Gebhardt J, Dasig D, Westphal LM, Behr B. Accuracy of day 3 criteria for selecting the best embryos. Fertil. Steril. 2002; 77:1191–1195.
Munné S. Chromosome abnormalities and their relationship to morphology and development of human embryos. Reprod. Biomed. Online 2006; 12:234–253.
Osada H, Aono F, Kuwayama M, Morita H, Teramoto S, Kato O. Clinical efficiency of vitrification on blastocysts transfer cycles. Fertil. Steril. 2003; 80:63.
Puissant F, Van Rysselberge M, Barlow P, Deweze J, Leroy F. Embryo scoring as a prognostic tool in IVF treatment. Hum. Reprod. 1987; 2:705–708.
Ren X, Liu Q, Chen W, Zhu G, Li Y, Jin L, et al.. Selection and vitrification of embryos with a poor morphological score: A proposal to avoid embryo wastage. J. Huazhong Univ. Sci. Technol. [Medical Sci. 2012; 32:405–409.
Ryan GL, Sparks AET, Sipe CS, Syrop CH, Dokras A, Van Voorhis BJ. A mandatory single blastocyst transfer policy with educational campaign in a United States IVF program reduces multiple gestation rates without sacrificing pregnancy rates. Fertil. Steril. 2007; 88:354–360.
Scott RT, Treff NR. Assessing the reproductive competence of individual embryos: a proposal for the validation of new “-omics” technologies. Fertil. Steril. 2010; 94:791–794.
Shaw-Jackson C, Bertrand E, Becker B, Colin J, Beaudoin-Chabot C, Rozenberg S, et al.. Vitrification of blastocysts derived from fair to poor quality cleavage stage embryos can produce high pregnancy rates after warming. J. Assist. Reprod. Genet. 2013; 30:1035–1042.
Shulman A, Ben-Nun I, Ghetler Y, Kaneti H, Shilon M, Beyth Y. Relationship between embryo morphology and implantation rate after in vitro fertilization treatment in conception cycles. Fertil. Steril. 1993; 60:123–126.
Stehlik E, Stehlik J, Paul Katayama K, Kuwayama M, Jambor V, Brohammer R, et al.. Vitrification demonstrates significant improvement versus slow freezing of human blastocysts. Reprod. Biomed. Online 2005; 11:53–57.
Styer AK, Wright DL, Wolkovich AM, Veiga C, Toth TL. Single-blastocyst transfer decreases twin gestation without affecting pregnancy outcome. Fertil. Steril. 2008; 89:1702–1708.
Zhu D, Zhang J, Cao S, Zhang J, Heng BC, Huang M, et al.. Vitrified-warmed blastocyst transfer cycles yield higher pregnancy and implantation rates compared with fresh blastocyst transfer cycles—time for a new embryo transfer strategy? Fertil. Steril. 2011; 95:1691–1695.
Ziebe S, Petersen K, Lindenberg S, Andersen AG, Gabrielsen A, Andersen AN. Embryo morphology or cleavage stage: how to select the best embryos for transfer after in-vitro fertilization. Hum. Reprod. 1997; 12:1545–1549.